Evaluating the Safety and Efficacy of Glucommander, a Computer-based Insulin Infusion Method, in Management of Diabetic Ketoacidosis in Children, and Comparing its Clinical Performance With Manually Titrated Insulin Infusion
Publication
Journal of Pediatric Endocrinology and Metabolism
Date
February 2009
Authors
Amber Fort,1 Umesh Narsinghani,2 Frank Bowyer1
Abstract
The Glucommander is a computer-based system for directing intravenous insulin infusion. Using a physician-selected glucose target range and a weight-based multiplier, it recommends an insulin infusion rate and interval to next glucose measurement. We evaluated the safety and efficacy of this system by conducting a retrospective chart review of 65 new-onset or existing type I diabetic children, admitted with diabetic ketoacidosis (DKA), managed with the Glucommander. We compared outcomes with 22 patients managed using manually titrated infusion. Time to glycemic control and correction of acidosis, number of insulin units used per kilogram per hour, and length of pediatric intensive care unit (PICU) and total hospital stay were analyzed using measures of central tendency. Children managed with Glucommander achieved equally rapid glycemic control and correction of acidosis, used less intravenous insulin, and spent less time in both PICU and hospital overall, compared to those managed with manual insulin infusion.
Introduction
Diabetic ketoacidosis (DKA) is a serious complication of diabetes caused by insulin deficiency and/or ineffectiveness. DKA occurs in 20-40% of children with new-onset diabetes mellitus. and in children with existing diabetes who develop infection or who miss insulin doses.1 DKA is a true medical emergency, carrying significant risk of mortality, and the pediatric patient has several unique characteristics that should be considered in management.2
Although intravenous fluid (IVF) alone in DKA can help decrease elevated blood glucose concentration,3 insulin therapy is essential to normalize blood glucose and correct ketoacidosis. Intravenous therapy must be titrated to adapt to the changing fluid. electrolyte, and dextrose needs of the patient because of dynamic. rapid intracellular shifting of molecules. This process requires close intensive care unit observation. Several methods have been used to achieve glycemic control and rehydration and are well-documented in the literature.1-4 Published protocols combine insulin infusion with maintenance IVF, and choose fluids based upon the patient’s initial electrolyte values. Manual titration of fluids and insulin, with multiple, sequential fluid bag changes based upon written scales determined by the physician, is commonly referred to in clinical practice as the “two-bag” method.5,6
The Glucommander is a computer-based system for controlling blood glucose by directing intravenous insulin infusion using bedside blood glucose measurement7 Davidson and Steed developed this system in 1984. Initial glucose target ranges along with a multiplier based upon the patient’s weight are selected, and the Glucommander then recommends an insulin infusion rate and a time to check the next blood glucose. The computer offers new recommendations hourly based upon the bedside glucose measurements, and adjusts the multipliers up or down depending on the patient’s proximity to the target. Glucommander algorithms arc programmed by the formula described by Davidson, Steed, and BoJe7 in which insulin dose/h = (blood glucose) – 60 x multiplier. The usual multiplier for an adult is 0.02. The initial multiplier is modified for the pediatric patient by the formula: weight in kilograms x 0.0002. The multiplier selected is only used by the computer to determine the initial insulin infusion rate. With serial entry of blood sugar results the computer algorithm adjusts the multiplier (and hence the insulin infusion rate) every hour based on the initially selected target blood glucose levels. We used a target blood sugar of 80-140 mg/dl. If the latest blood sugar entered is in target range the computer algorithm does not change the multiplier. If the blood sugar is less than target the multiplier is decreased by 50%. If the blood sugar is still above target and has not decreased from the previous value by 25% the multiplier is increased by 50%.7 This allows blood sugar decrements of up to 100 mg/dl.
The Glucommander has been extensively studied and accepted as a standardized treatment method applicable to a wide variety of conditions in adults, including ketoacidosis, hyperosmolar non ketotic states, gastroparesis, gastroenteritis, pancreatitis, peri- and post-operative glucose management, steroids-induced hyperglycemia, labor, sepsis, and for establishing the degree of insulin sensitivity.5 It is a simple, safe, and effective method of achieving glycemic control, and has been shown to obviate the need for variable schedule glucose management, wasted IVF from multiple fluid bag changes, and prolonged hospital and intensive care unit stays, all associated with higher costs. However, there is limited information regarding its use in the pediatric population. We now report our experience with the Glucommander in the management of pediatric DKA, compared to the outcomes achieved using a two-bag protocol. We hypothesized that use of the Glucommander in children would achieve the same advantages seen in adults. We compared time required to achieve glycemic control, time required to correct acidosis, total intravenous insulin requirement, and hospital and PICU length of stay using the Glucommander with outcomes achieved using a two-bag, manually titrated method of insulin infusion.
Methods
A retrospective chart review was conducted of all new-onset or existing type I diabetic children admitted to the pediatric intensive care unit (PICU) between January 2004 and October 2006 with DKA. The children ranged in age from 2-18 years (mean 12 years). Parameters for assessment were determined prior to chart review. Basic demographics, including race, gender, age at presentation, and age at diagnosis of type 1 diabetes, were noted (Table 1). The starting points for analysis were the blood glucose and serum bicarbonate values obtained at presentation to our facility, even for children transported from outlying emergency rooms who had already received insulin and fluids.
Number of hours to glycemic control, defined by first blood glucose level <200 mg/dl, and correction of acidosis, defined by first serum bicarbonate >16 mmol/l, were recorded. The number of intravenous insulin units per kilogram per hour was evaluated, as well as total hospital and PICU length of stay in hours. Any subcutaneous insulin administration of any insulin type, whether administered simultaneously with intravenous infusion or not, was not included in the total number of insulin units received. Admissions were also reviewed for two major complications: inadvertent hypoglycemia, defined as blood glucose less than 40 mg/dl, or cerebral edema.
Of 90 identified charts, three were excluded from the study because the patient’s condition was effectively managed with only subcutaneous rather than intravenous insulin infusion. Of the 87 patient charts selected for review, 65 had been managed using the Glucommander and 22 had been managed using the two-bag method. A subset of six children had experienced multiple admissions for DKA, and had been managed with both methods of insulin infusion over the time period selected. These children represented a unique opportunity to compare outcomes in the absence of confounding demographic factors. The Glucommander was introduced into our hospital in the adult endocrine service several years ago as the standard for managing hyperglycemic conditions. Based on their success the pediatric endocrine service revised the adult protocols for use in the pediatric patient and began its use in the Children’s Hospital. The Glucommander gradually evolved as the protocol of choice in our unit over the time period selected for review a!: the pediatric intensive care and emergency room teams became more familiar with it. Informed consent for use of the Glucommander was not obtained for any child, because the basis for its use is still intravenous insulin infusion, and did not require deviation from the recommended standard of care for management of DKA. This study then compared the group of children treated since the introduction of the Glucommander protocol with a group of patients treated with the two-bag method which had been the protocol of choice prior to the introduction of the Glucommander.
Data were summarized using statistical measures of central tendency. The main assumption for analysis is that the Glucommander and two-bag groups represent random samples from the same larger population to which we wish to apply our conclusions. Data were summarized using statistical measures of central tendency then means observed in the Glucommander and the two-bag groups were compared using the unpaired t-test.
Table 1. Demographic characteristics of children with type 1 diabetes mellitus admitted for management of diabetic ketoacidosis using the Glucommander vs manually titrated infusion
| Glucommander | Manual Infusion | |
|---|---|---|
| Average Age (yr) | 12 | 12 |
| Average Age at Diagnosis (yr) | 9 | 8 |
| Race (%) | 85% Black 15% White |
86% Black 14% White |
| Gender (%) | 46% Male 37% Male |
54% Female 63% Female |
| Average Weight (kg) | 51 | 40 |
Results
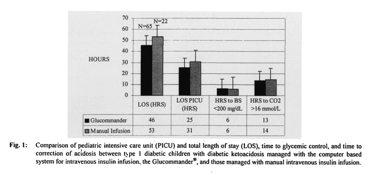
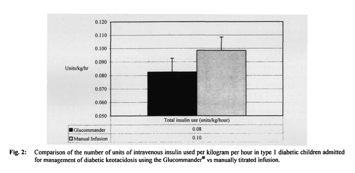
In children managed with the Glucommander, the mean blood glucose at presentation was 464 mg/dl, and mean serum bicarbonate was 11 mmol/l. Mean times to glycemic control and the correction of acidosis were 6 (± 5) hours and 13 (± 10) hours, respectively; mean intravenous insulin use was 0.08 (± 0.08) U/kg/h. Mean PICU LOS was 25 (± 12) hours; total LOS 46 (± 28) hours.
In children managed with the two-bag method, the mean blood glucose at presentation was 410 mg/dl, and mean scrum bicarbonate was 9 mmol/l. Mean times to glycemic control and correction of acidosis were 6 (± 5) and 14 (± 11) hours, respectively. Mean insulin use was 0.10 (± 0.11) U/kg/h. Mean PICU LOS was 31 (± 9) hours; total LOS 53 (± 21) hours (Fig. 1). No incidents of inadvertent hypoglycemia (blood glucose < 40 mg/dl) or cerebral edema were noted for any child, regardless of the management method employed.
Children managed with the Glucommander had an observed reduction in the number of insulin units used per kilogram per hour (p <0.05), and reduction in length of both overall hospital (p <0.04) and PICU stay (p <0.09) compared to the two-bag method (Figs. 1, 2). There was no clinically significant difference in the number of hours to glycemic control (p <0.06) and a minimal decrease in time to correction of fluid deficit using the Glucommander versus the two-bag method (p <0.09).
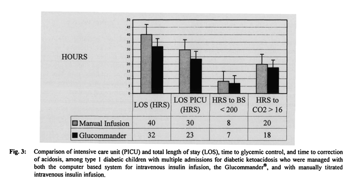
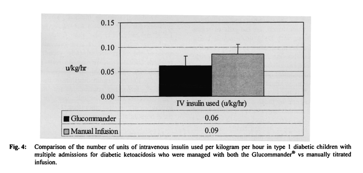
Among the subset of children with multiple admissions for DKA who had been managed with both methods, use of Glucommander resulted in shorter mean hospital (32 ± 11 vs 40 ± 15 h) and PICU stays (23 ± 10 vs 30 ± 11 h), use of less insulin (0.06 ± 0.02 U/kg/h vs 0.09 ± 0.10 U/kg/h), faster glycemic control (7 ± 5 vs 8 ± 5 h) and faster correction of acidosis (18 ± 11 vs 20 ± 12 h) compared to the two-bag method (Figs. 3, 4). The small number of children in this subpopulation suggests that this difference is not clinically significant.
Discussion
The Glucommander compared favorably with results obtained using existing protocols for DKA management in the literature, showing that the Glucommander is safe and effective in allowing timely achievement of glycemic control, correction of acidosis, and smooth transition to home regimens. It is possible that the smaller number of patients in the two-bag group compared to the Glucommander group influenced the assessed parameters. However, the results from that group were consistent with our previous clinical experience managing DKA and arc therefore representative.
It is noteworthy that the majority of children presenting to our facility for management of DKA are transported from outlying emergency rooms and/or physician offices, and most had received insulin and fluids prior to presentation in variable quantities and with variable effectiveness. Additionally, the impetus for DKA in individual children. including whether or not they were ill, had been non-compliant, or experienced trauma, was not considered, and could have impacted not only choice of fluids and insulin therapy but how quickly clinical resolution was .obtained. However, these variables are true for both groups of patients, and our study is felt to represent the population of patients that present to a PICU for management.
Our results suggest that the Glucommander is as safe and effective as other traditional methods of IV insulin infusion in children. There was no negative impact upon patient safety, including complications of inadvertent hypoglycemia or cerebral edema. Several other important clinical observations were made in the group managed with the Glucommander: nursing satisfaction and labor intensiveness on the part of the PICU team favored the Glucommander; it was perceived as user friendly by house staff and pharmacy personnel when compared to the two-bag method. These findings are opinion-based, on the part of teams caring for children with DKA on a regular basis, and have not yet been subjected to rigorous study. The system is also intuitively less error-prone because it is computer-based rather than written scale based, and requires only a few written orders to initiate. The most significant limitation of the Glucommander is the fact that there is a learning curve to the program by those using it. It also still requires hourly manual finger-sticks. Careful initial data entry, such as correct target glucose levels and correct initial multiplier section based on patient weight, is required to avoid incorrect insulin infusion rates by the computer algorithm. Importantly, it does not diminish the need for appropriate fluid and electrolyte management in critically ill children with DKA.
Several other unexpected advantages were revealed using the Glucommander. First, the protocol can be initiated and easily managed in the emergency department. allowing immediate access to controlled IV insulin infusion regardless of bed limitations in the PICU, and second, a cost savings of US $600 per patient per PICU day was achieved by using the Glucommander due to shorter length of stay.
Affiliations
- Department of Pediatrics, Mercer University School of Medicine, Macon, Georgia.
- Division of Pediatric Intensive Care, Mercer University School of Medicine, Macon, Georgia.
Acknowledgments
The authors wish to thank the medical records department at the Medical Center of Central Georgia for their assistance in compiling charts for review; Cynthia Loudin, NPC, and Jennifer Campbell, NPC, for assistance in data collection and direct patient care; Jeannie Li, MS3, for assistance in data collection; Rogelio dcla Cruz, MD, for the study idea, direct patient care and editing assistance; and pediatric house staff, pediatric critical care nursing staff, and pharmacy personnel for providing subjective input regarding the Glucommander.
Disclosures
The authors have nothing to disclose in regard to affiliations with, or financial interests in, any organization that may have an interest in any part of this study.
References
- Klekamp J, Churchwell KB. Diabetic ketoacidosis in children: initial clinical assessment and treatment. Pediatr Ann 1999; 25: 387-393.
- Walsdorf J, Glaser N, Sperling MA. Diabetic keto acidosis in infants, children, and adolescents: a consen sus statement from the American Diabetes Association. Diabetes Care 2006; 29: 1150-1159.
- Alemzadeh R, Wyatt DT. Diabetes mellitus in children. In: Behrman RE, Kliegman RM, Jenson HB, eds. Nelson Textbook of Pediatrics, I 71h Ed. Philadelphia, PA: Saunders, 2004; 1947-1972.
- American Diabetes Association. Position statement: Hyperglycemic crises in patients with diabetes mellitus. Clin Diabetes 200 I; 19: 82-90.
- Grimberg A, Cerri R, Satin-Smith M, Cohen P. The “two bag system” for variable intravenous dextrose and fluid administration: benefits in diabetic ketoacidosis management J Pediatr 1999; 134: 376-378.
- Poirier M, Greer D, Satin-Smith M. A prospective study of the “two-bag system” in diabetic ketoacidosis management. Clin Pediatr 2004; 43: 809-813.
- Davidson PC, Steed RD, Bode BW. Glucommander: a computer-directed intravenous insulin system shown to be safe, simple, and effective in 120,618 h of operation. Diabetes Care 2005; 28: 2418-2423.