Presentation
International Hospital Diabetes Meeting
Date
May 2015
Authors
Joseph Aloi, Paul Chidester, Amy Henderson, Raymie McFarland, Robby Booth, Melanie Mabrey
Objective
Patients undergoing cardiovascular (CV) surgery and requiring intravenous (IV) insulin infusions challenge the clinician with achieving CMS measures that are changing over time. This study was intended to evaluate the efficiancy and safety using Glucommander (GM) to achieve target blood glucose different measurement timelines.
Methods
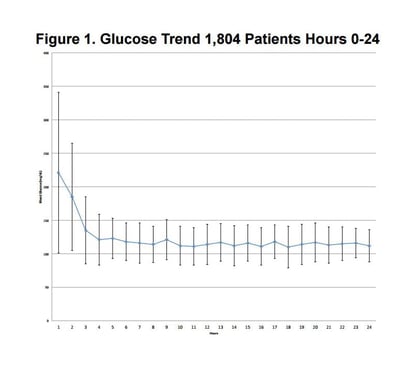
1,804 patients undergoing CV surgery were evaluated. Glucose control was analyzed over time on Glucommander IV (GM IV) and Subcutaneous (GM SubQ) insulin therapy. Qualifying patients were started on GM IV insulin and transitioned to GM SubQ (179 Patients) if ongoing therapy was deemed necessary. The efficacy and safety of IV and SubQ were evaluated by end anesthesia time intervals: (1) Blood Glucose (BG) Average at 12-24 and 12-72 hours, (2) Percent in target <180 mg/dL, (3) Hypoglycemic events <40 and <70 mg/dL, and (4) Average BG for subgroup who received meals during IV treatment.
Results
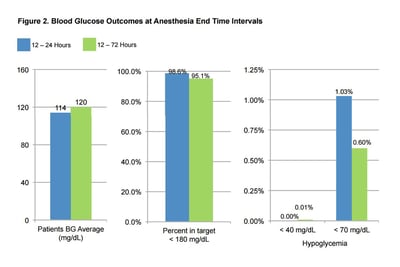
BG average for patients 12-24 hours was 114 mg/dL and 120 mg/dL for 12-72 hours. Percent in target <180 mg/dL was 98.6% for 12-24 hours and 95.1% for 12-72 hours. Hypoglycemia defined as <40 mg/dL or <70 mg/dL was 0.0% and 1.03% respectively for hours 12-24; for the broader time range of 12-72 hours, hypoglycemia was 0.01% and 0.6% respectively. A total of 1,010 carbohydrate-containing meals were recorded on GM IV with an average meal BG of 122.7 mg/dL.
Conclusion
Patients using GM IV and SubQ achieve BG values within the prescribed target range regardless of measurement timeframe, with a very low incidence of hypoglycemia. These results suggest that GM IV and SubQ can safely maintain glucose targets with very minimal risk of hypoglycemia for CV surgery patients, even those who also receive oral calorie intake.