Presentation
American Association of Clinical Endocrinologists Scientific & Clinical Congress
American Diabetes Association Scientific Sessions
Date
May 2014
June 2014
Authors
Joseph Aloi,1 Paul Chidester,2 Raymie McFarland3
Objective
Hospitalized patients who experience hyper/hypoglycemia have been associated with increased mortality and hospital cost as compared with patients with an absence of hypoglycemia. Subcutaneous insulin titration to target glucose levels has always been a difficult task. Sentara’s successful experience with Glucommander (GM), a web-based insulin algorithm integrated with the hospital EHR at Virginia Beach General Hospital for IV inpatient insulin protocols suggested that its expanded use across the Sentara Health System may improve glycemic control for Subcutaneously (SQ) treated patients with Basal Bolus Insulin (BBI) placed through CPOE. We studied the glycemic impact using the GM SQ system for treatment of patients with hyperglycemia compared to BBI therapy alone.
Methods
This 30-day quality initiative is being conducted at Sentara Healthcare System in southeast Virginia across 2 Hospital sites. Patients needing SQ insulin were placed on the GM system and we measured the first 30 days of use to the hypoglycemia baseline of the 30 days prior to GM at each site. BBI placed by CPOE was used to treat insulin patients during the baseline period. The glycemic targets being used for this project were % of blood glucose events <70 mg/dl and Glucose Profile Day 1 vs Day of Discharge for the 178 patients in this analysis.
Results
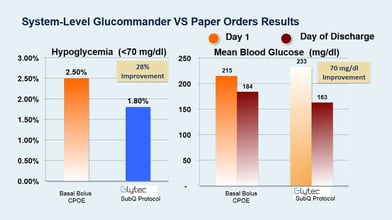
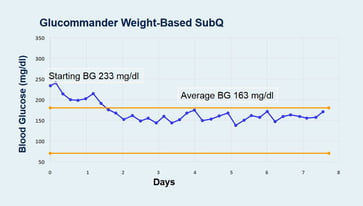
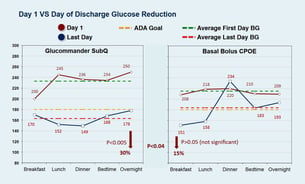
Baseline hypoglycemia episodes (<70 mg/dl) were reduced by 28% (P <0.05) with GM SQ compared to BBI. GM SQ had a Day 1 to Day of Discharge delta of 70 mg/dl from 233 to 163 mg/dl for a 30% reduction (P <0.005). BBI went from 215 to 184 (P >0.05) for a 15% reduction. GM SQ patients had average of 2x glucose reduction from baseline compared to patients treated with BBI (P <0.04). Day of Discharge glucose average for GM SQ was 163, and coincides with the midpoint of the prescribed target range of 140-180 (mg/dl).
Subcutaneous Insulin Therapy on the Gen/Med Floors over a 30-day Period
| Patients | Glucommander SubQ BBI Orders | Non-Glucommander BBI CPOE |
|---|---|---|
| Average Initial Blood Glucose (BG) | 233 mg/dl | 215 mg/dl |
| Average Glucose Day of Discharge | 163 mg/dl | 184 mg/dl |
| Average Glucose Reduction | 70 mg/dl | 31 mg/dl |
| % Average Glucose Reduction | 30% | 15% |
| Hypoglycemia <70 mg/dl | 1.8% (43 BGs) | 2.5% (125 BGs) |
| Hypoglycemia <40 mg/dl | 0.0% (0 BGs) | 0.4% (21 BGs) |
Conclusion
Use of the eGlycemic Management System GM SQ to treat hyperglycemic patients on SQ insulin showed dramatic reductions of up to 28% in hypoglycemia in the first 30 days of use and 2x the hyperglycemic improvement compared to BBI treatment orders. Our results suggest that GM SQ is a safe and clinically effective tool for improving glycemic control for patients who need SQ treatment while in the hospital and showed statistically significant reductions in daily glucose compared to standard basal bolus orders.
Affiliations/References
- Eastern Virginia Medical School, Norfolk, Virginia.
- Sentara Healthcare, Norfolk, Virginia.
- Glytec, Greenville, South Carolina.